In the last article, we discussed quenching of aluminum, and the importance of rapid quenching to prevent the formation of precipitates at the grain boundaries. Water is often used as a quenchant but can cause high residual stresses and distortion.
To eliminate or reduce distortion and minimize residual stresses, many companies have used polyalkylene glycol to dramatically reduce distortion and residual stresses.
Polyalkylene Glycol
Polyalkylene glycol (PAG) quenchants are the most used polymer quenchants on the heat-treating market today. Polyalkylene glycols (PAG), or polyalkylene glycol ethers, were first introduced as quenchants in the early 1970s. PAG quenchants are an example of a copolymer. This quenchant is derived from two monomeric units — ethylene oxide and propylene oxide — to form polyalkylene glycol (Figure 1).
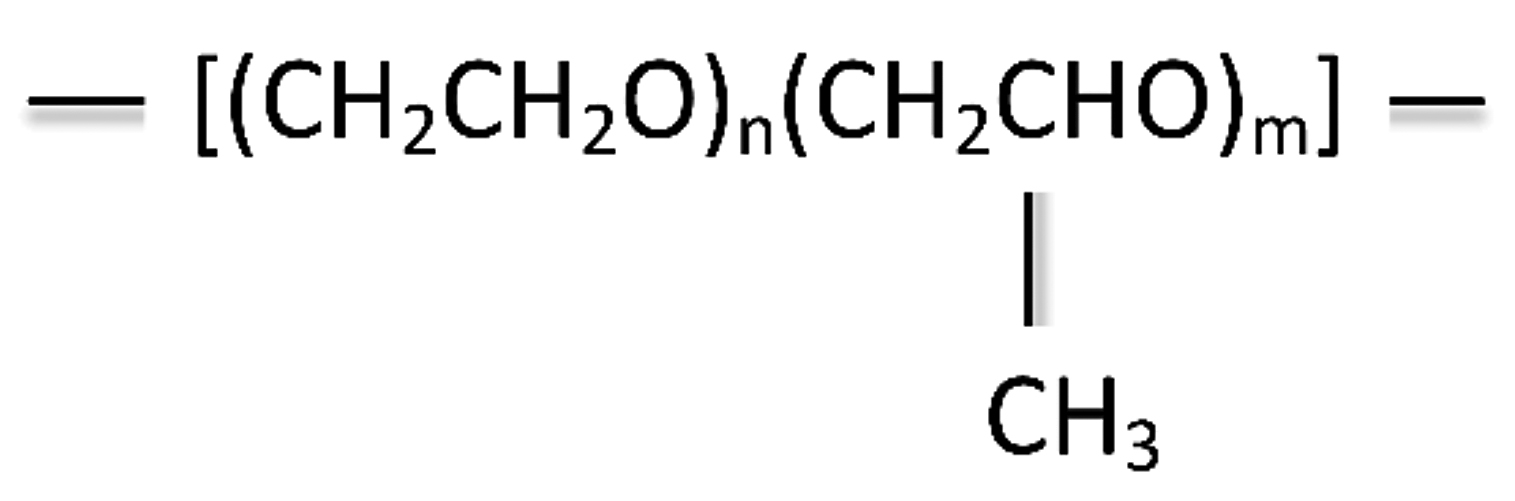
By varying the molecular weights and the ratio of oxides, polymers having broad applicability may be produced. Proper selection of the polymer composition, and its molecular weight, provides a PAG product that is completely soluble in water at room temperature.
PAG quenchants are used widely as an alternative to water or oil for the quenching of aluminum parts such as thin-section airframe and skin components, castings and extrusions for aerospace applications, and engine blocks, cylinder heads, and wheels for the automotive industry.
Quenching speed is critical to prevent the precipitation of intermetallic compounds which would have an adverse effect on mechanical properties and corrosion resistance.
The controlled uniform quenching characteristics of PAGs can significantly reduce or even eliminate the distortion often associated with water quenching without impairing mechanical properties or corrosion resistance. This is particularly important with thin-section sheet aluminum airframe components in the aerospace industry and complex castings and forgings, which are often quenched into boiling water or mineral oil to minimize distortion.
PAG quenchants are generally used at a concentration of 10-20 percent for heavy-section castings and forgings, and at 25-40 percent for thin-section sheet components. Figure 2 shows the effect of concentration on the cooling curves of a typical PAG quenchant.
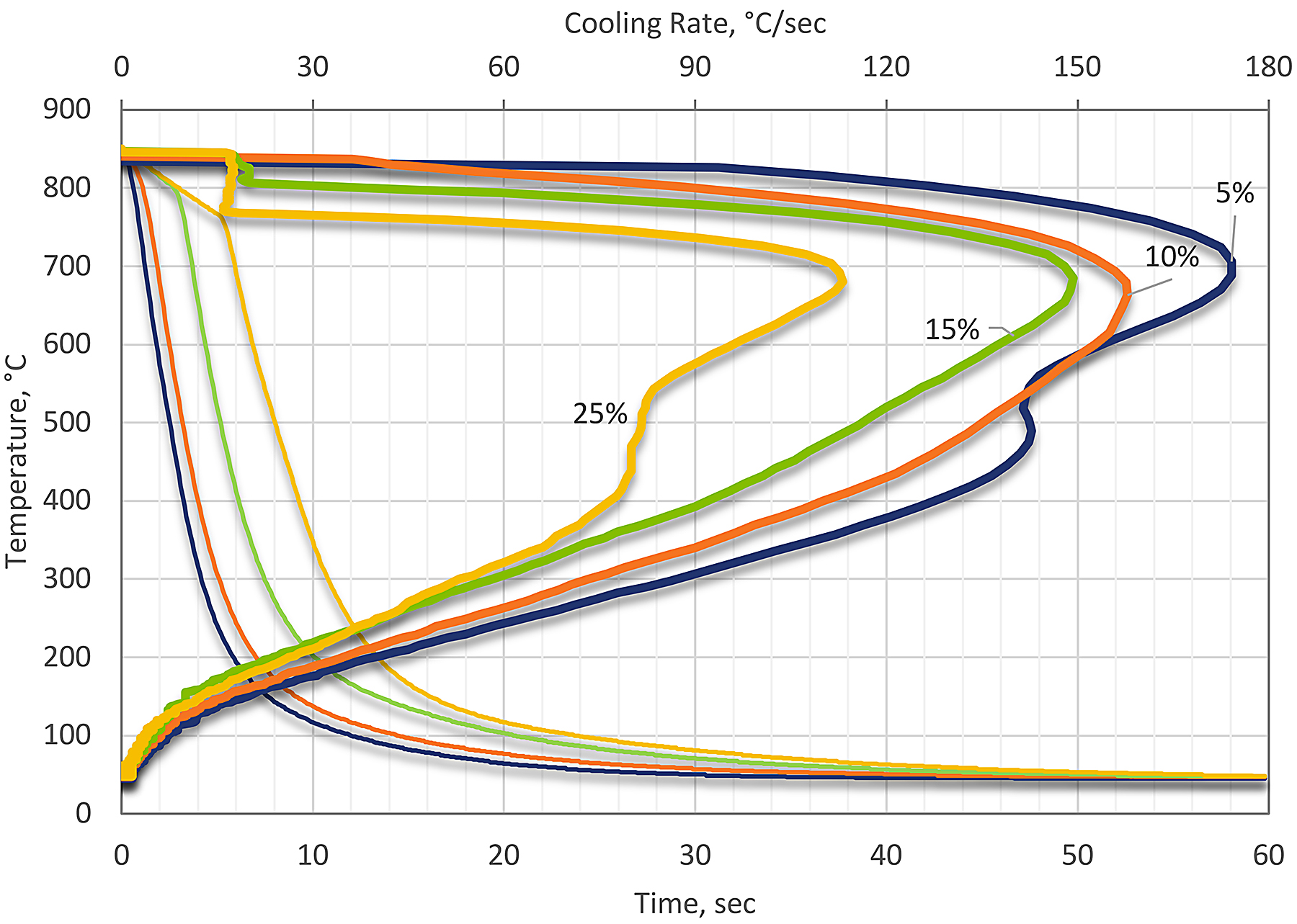
PAG quenchants can be used in conjunction with both recirculating air furnaces and molten salt baths. However, with molten sat baths, special precautions must be taken to compensate for salt carry-over and to maintain the polymer solution at the correct working concentration.
These quenchants are available in a wide variety of viscosities and molecular weights. Commercial quenchants also contain additional additives such as corrosion inhibitors, defoamers, fungicides, and biocides to enhance performance in service.
Polyalkylene glycols exhibit inverse solubility in water [1]. They are completely soluble at room temperatures but become insoluble at elevated temperatures. This inverse solubility can range from 60°C to 90°C depending on the molecular weight of the polymer. This phenomenon of inverse solubility modifies the conventional three-stage quenching process and provides great flexibility in controlling cooling rate.
PAG quenchants are approved by major aerospace manufacturers worldwide and are used extensively for critical applications in aircraft manufacture (Figure 3).

Polyalkylene glycol polymer quenchants are used in the aerospace industry to control and minimize the distortion occurring during the quenching of aluminum. Typically, these quenchants are governed by AMS 3025 [3] and are either Type I or Type II quenchants. Type I quenchants are single polyalkylene glycol polymers, while Type II quenchants are multiple molecular weight polyalkylene glycol polymers. Each offers different benefits. Because of the higher molecular weight of the Type II PAG quenchants, lower concentrations can be used.
Application of polyalkylene quenchants, such as Aqua-Quench® 260, are effective in reducing the residual stresses (and distortion) after quenching. The effect of quenching aluminum on residual stresses in Aqua-Quench 260 is shown in Figure 4.
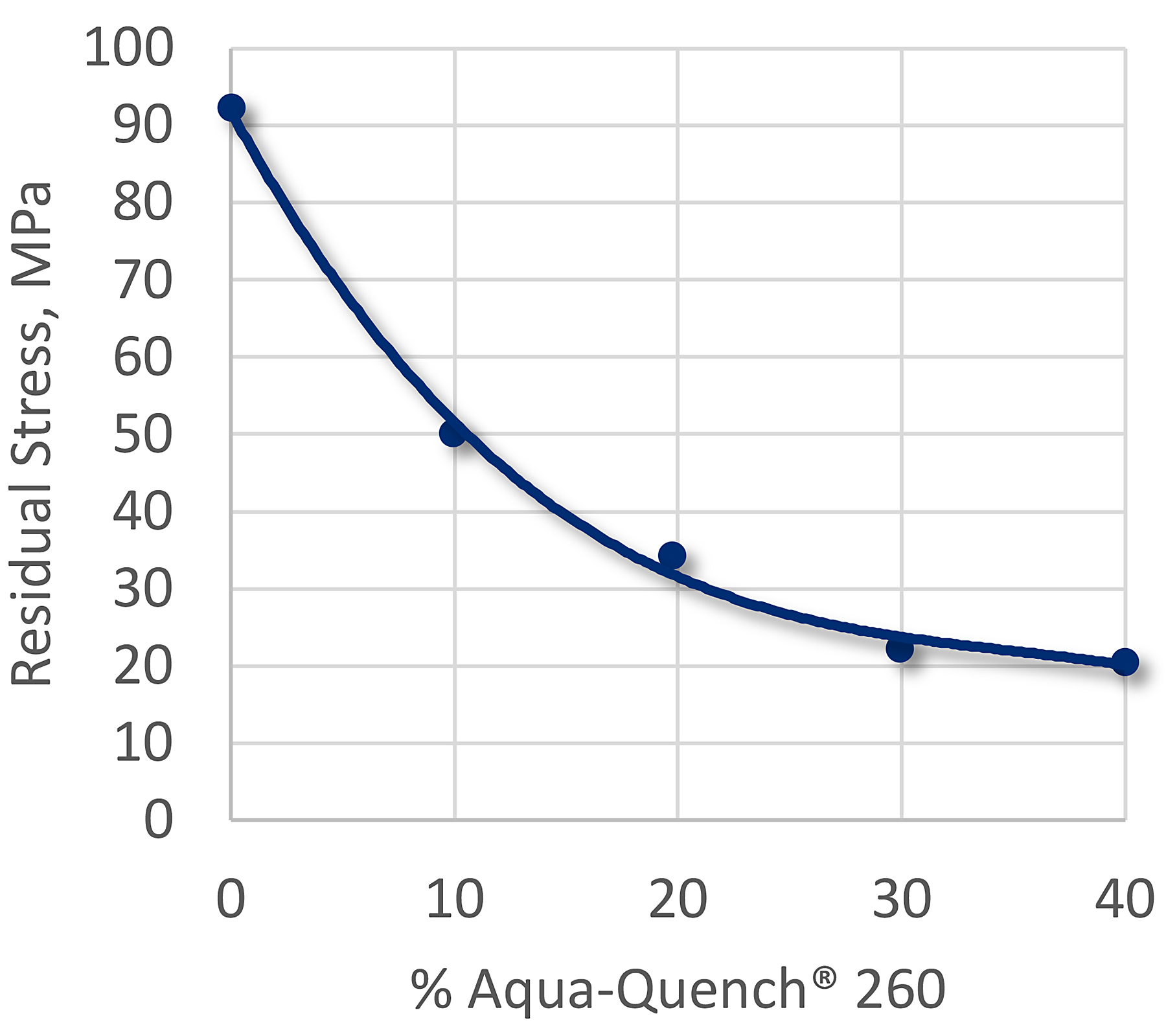
The concentration of the polymer influences the thickness of the polymer film that is deposited on the surface of the part during quenching. As the concentration increases, the maximum rate of cooling, and the cooling rate in the convection phase, decrease. As the concentration of PAG is increased, the maximum cooling rate also decreases. This is shown in Figure 5 for sheet metal, and for forgings and wrought plate.

Agitation has an important effect on the quenching characteristics of the polymer quenchant. It ensures uniform temperature distribution within the quench tank, and it also affects the quench rate. As the severity of agitation increases, the duration of the polymer-rich phase decreases and eventually disappears, and the maximum rate of cooling increases. Agitation has comparatively little effect on the cooling rate during the convection stage for polymer quenchants.
The refractive index of PAG polymer solutions (in the range employed for quenching) is essentially linear with concentration. Thus, the refractive index of a PAG quenchant solution serves as a measure of product concentration. Industrial model optical refractometers that employ an arbitrary scale may be calibrated. Whereas such instruments prove invaluable for day-to-day monitoring of the quenchant concentration, the refractometer also will register other water-soluble components that are introduced to the used quenchant. When the indicated refractometer reading begins to provide erroneous numbers, some other analytical test is required to define the “effective” quenchant concentration. With PAG quenchants, kinematic viscosity measurements (which are correlated with concentration) have proven to be most useful.
As required, additional analytical tests for pH, inhibitor level, and conductance may be useful adjuncts to a successful monitoring program. If the level of contaminants in the PAG quenchant becomes excessive-where these contaminants may be, in part, the same undesirable constituents that are detrimental to water alone, or oil-quenchant recovery can be affected thermally. By heating the quenchant solution (in whole or in part) above the separation temperature, a more-dense polymer-rich layer is obtained. Much of the water-soluble contamination can be withdrawn with the supernatant water layer. Solid contaminants such as scale or carbon would require settling, filtration, and/or centrifugation.
Because PAG quenchants are, for the most part, resistant to bacteria and fungus, the addition of a bactericide to the as-supplied quenchant is not required. Further, biochemical activity in use is traceable not to the PAG polymer itself but to the introduction of nutrient contaminants. Microbiological treatment such as is employed with other aqueous metal working fluids generally will keep under control this foreign biological activity.
Conclusions
In this short article, the benefits of the use of PAG quenchants in reducing distortion and residual stress was illustrated. Concentrations from 10-40 percent are used to quench aluminum sheet metal, extrusions, and forgings of all the heat treatable aluminum alloys to reduce distortion and subsequent check-and-straighten activities.
In the next article, we will discuss natural aging of aluminum and the various precipitation mechanisms that enable aluminum to harden to the desired strength after solution heat treatment and quenching. Should you have any suggestions for any articles or comments regarding this article, please contact the writer, or the editor.
References
- G. Totten, C. Bates and N. Clinton, Eds., Handbook of Quenching and Quenchants, Metals Park, OH: ASM International, 1993.
- P. M. Kavalco, L. C. Canale and G. E. Totten, “Distortion Reduction by Aqueous Polymer Quenching of Aluminum Alloys,” Industrial Heating, vol. 2, no. February, p. 39, 2011.
- SAE, “AMS 3025E Polyalkalene Glycol Heat Treat Quenchant,” SAE, Warren, PA, 2018.

























